Lentivirus titration
LabOmics offers 2 methods of lentiviral titration:
1. Quick immuno-based method: rapid lateral flow immunoassay that allows quick confirmation of useful lentivirus titers in the packaging supernatants. The test detects lentiviral capsule proteins released into the packaging supernatant as a surrogate marker to determine whether virus production yields a usable titer for subsequent infection. This assay takes only 10-15 minutes, compared to the conventional re-infection based assay that takes 2-3 days.
2. Lenti-Pac™ HIV and FIV qRT-PCR Lentivirus Titration Kits, designed for quick and simple titration of lentiviral particles to determine the virus copy numbers and to confirm the success of packaging reactions. The kits include the RNA extraction reagent, qRT-PCR reagents and standard control. The RNA genome of lentivirus is reverse-transcribed and quantified using qRT-PCR and SYBR® green technologies.
-
 Lenti-Pac™ HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions)440,00 €Lenti-Pac™ HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions) Learn More
Lenti-Pac™ HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions)440,00 €Lenti-Pac™ HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions) Learn More -
 Lenti-Pac™ HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions)596,00 €Lenti-Pac™ HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions) Learn More
Lenti-Pac™ HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions)596,00 €Lenti-Pac™ HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions) Learn More -
 Lentivirus Titration XpressCard (6 tests)155,00 €Rapid Lentivirus Titration XpressCard - 6 tests Learn More
Lentivirus Titration XpressCard (6 tests)155,00 €Rapid Lentivirus Titration XpressCard - 6 tests Learn More -
 Lentivirus Titration XpressCard (10 tests)210,00 €Rapid Lentivirus Titration XpressCard - 10 tests Learn More
Lentivirus Titration XpressCard (10 tests)210,00 €Rapid Lentivirus Titration XpressCard - 10 tests Learn More -
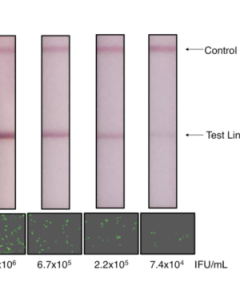 Lentivirus Titration XpressCard (20 tests)350,00 €Rapid Lentivirus Titration XpressCard - 20 tests Learn More
Lentivirus Titration XpressCard (20 tests)350,00 €Rapid Lentivirus Titration XpressCard - 20 tests Learn More -
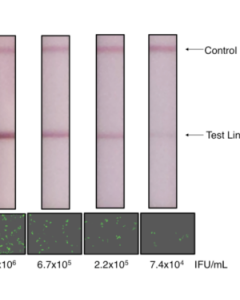 Lentivirus Titration XpressCard (50 tests)750,00 €Rapid Lentivirus Titration XpressCard - 50 tests Learn More
Lentivirus Titration XpressCard (50 tests)750,00 €Rapid Lentivirus Titration XpressCard - 50 tests Learn More -
 Lenti-Pac HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions)400,00 €Lenti-Pac HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions) Learn More
Lenti-Pac HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions)400,00 €Lenti-Pac HIV qRT-PCR Titration Kit (20 RT reactions, 50 PCR reactions) Learn More -
 Lenti-Pac HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions)550,00 €Lenti-Pac HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions) Learn More
Lenti-Pac HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions)550,00 €Lenti-Pac HIV qRT-PCR Titration Kit (40 RT reactions, 100 PCR reactions) Learn More
